
Unmyelinated Aortic Baroreceptor Activity is Reduced in a Rabbit Model of Atherosclerosis
HypertensionReceived 02 Apr 2026 Accepted 27 Apr 2026 Published online 29 Apr 2026
ISSN: 2995-8067 | Quick Google Scholar
END
Submit Article


Received 02 Apr 2026 Accepted 27 Apr 2026 Published online 29 Apr 2026
Aims: Arterial baroreceptors are located in vessels which are subject to atherosclerotic changes. Using a rabbit model, we investigated effects of atherosclerosis on arterial baroreceptors, arterial baroreflex regulation and control of Mean Arterial Pressure (MAP).
Methods and results: Seven-week-old male New Zealand White rabbits were placed on an atherogenic diet for 14 weeks (treatment). Treatment produced significant atherosclerosis along the aorta, but spared the carotid bifurcation. Treated animals had higher MAP: conscious–(treated: 80.1 [77.3, 82.9] versus control: 70.9 [69.9, 71.9] mmHg; N = 7 and 9 respectively; p = < 0.001) (mean [90% confidence interval]), anaesthetized–(treated: 99.9 [97.2, 102.7] versus control: 95.6 [93.5, 97.7] mmHg, N = 24 for each group; p = 0.050). In anaesthetized animals Pulse Wave Velocity (PWV) along the aorta was higher in treated animals (5.64 [5.54, 5.74] versus 5.07 [4.98, 5.16] m/s in control, N = 24 for each group; p < 0.0001). MAP was ramped (from 40 to 140 mmHg) using inflatable cuffs around the descending aorta and posterior vena cava. Treatment did not affect the activity of individual A-fibre baroreceptors in the ADN. Multifibre records of activity in the left Aortic Depressor Nerve (ADN) and left Carotid Sinus Nerve (CSN), showed that treatment reduced C-fibre baroreceptor activity in the ADN; there was ~16.5% less integrated activity at MAP >100 mmHg in treated animals (4650 [4174, 4988] versus 5570 [5220, 5919] au in control; N = 22 and 20, respectively; p < 0.001). Treatment had no effect on multifibre recordings from the CSN.
In conscious animals, baroreflex curves describing control of Renal Sympathetic Activity (RSNA) were obtained using i.v. phenylephrine and nitroprusside to modify MAP. Treatment shifted the mid-point of the curve to a higher MAP (72.4 [69.9, 74.8] versus 63.6 [61.3, 65.8] mmHg in control; N = 5 and 8, respectively; p = 0.02. In anaesthetized animals, baroreflex curves for RSNA were obtained using inflatable cuffs to control MAP. When input from the CSN was kept low and steady by common carotid artery occlusion, a high level of MAP was unable to fully inhibit RSNA (12.5 [8.4, 16.6] and 3.9 ([2.6, 5.2] n.u. at saturation in 5 treated and 7 control animals, respectively; p = 0.01).
Conclusion: The loss of aortic C-fibre baroreceptor activity that occurs in this model of atherosclerosis may reset MAP to a higher value, since C-fibre input is likely a controller of MAP.
Atherosclerosis is a chronic progressive inflammatory disease that affects the walls of larger systemic arteries (> 0.2 mm diameter), affecting both conduit and muscular arteries []. It involves gradual stiffening of the artery with the development of plaques in the intimal layer of the artery through uptake and incorporation of lipids into the arterial wall, fibrosis, and eventual calcification of the plaques.
Risk factors for atherosclerosis include inflammation, elevated circulating levels of cholesterol (low density lipoproteins) and triglyceride (and high saturated fat diet), elevated blood pressure (hypertension), smoking, Types 1 and 2 diabetes, obesity (and metabolic syndrome), and physical inactivity [2,3]. Risk factors are identified based on the coincidence/correlation of the risk factor with the incidence of atherosclerosis. Current thinking is that dysfunction of the endothelium with inflammation and elevated blood lipids is necessary for initiation and development of atherosclerosis [4]. While hypertension may not be present in all individuals suffering atherosclerosis–and is therefore not essential for the development of the disease–its presence appears to promote and accelerate the disease process. It has become clear that even very modest elevations in arterial pressure may contribute to the development of atherosclerosis [5-7]. Atherosclerotic lesions rarely occur in systemic veins or in the pulmonary circulation, even when these vessels are exposed to the same inflammatory and blood lipid profiles as systemic arteries. However, when they are subject to unusually high pressure, they too may develop atherosclerotic lesions; atherosclerosis has been observed in saphenous veins used as graft vessels in coronary artery bypass surgery [8] and in pulmonary arteries of individuals suffering from pulmonary hypertension [9,10].
The association of atherosclerosis and hypertension may also reflect reverse causation, meaning that while hypertension can promote atherosclerosis, the reverse can occur–that atherosclerosis may promote hypertension. An example of this is the secondary hypertension that develops when atherosclerotic lesions cause severe narrowing of the renal arteries [11].
Systemic Arterial Pressure (AP) is normally tightly regulated and, to a lesser extent, controlled by the arterial baroreceptor reflex. While the primary role of the baroreflex lies in the moment-to-moment regulation of arterial pressure [12], there is evidence that it may also play a role in the long-term setting of the absolute value of AP [13-15]. Sensory components of this reflex, the arterial baroreceptors, are located in conduit arteries (aortic arch, the origin of the subclavian artery, and the carotid sinus). Baroreceptors respond to the distortion of the vessel wall that accompanies each pressure pulse. Vessels bearing baroreceptors are affected early in the development of atherosclerosis, and atherosclerotic changes–increased vessel wall stiffness and the production of inflammatory products and free radicals–may reduce baroreceptor input at any given pressure [16]. In humans, reflex baroreflex control of HR is reduced in individuals suffering from atherosclerosis [17,18]. Three studies have investigated the effect of atherosclerosis on arterial baroreflex function in animal models. (hyperlipidemic greyhound model of atherosclerosis [19], the Watanabe Heritable Hyperlipidemic (WHHL) rabbit model of atherosclerosis [20,21]. These studies found that atherosclerotic lesions in the carotid sinus and aortic arch were associated with reduced ability of the baroreflex to regulate arterial pressure, and control heart rate and Renal Sympathetic Nerve Activity (RSNA). In each paper, it was implied that reduced baroreflex function was a consequence of reduced responsiveness of arterial baroreceptors. Whether other components of the arterial baroreflex–central and autonomic nervous control of effector mechanisms (heart rate and contractility; arterial resistance and capacitance)–are affected by atherosclerosis has not yet been established (for example, [22]), but, clearly, arterial baroreceptor input is an important component affected by atherosclerosis.
Angell-James (1974) examined aortic baroreceptor activity in rabbits fed a diet designed to produce atherosclerosis (1% cholesterol, 6% sunflower oil for up to 67 weeks) [23]. Compared with control, there was lower aortic arch distensibility in animals with atherosclerotic lesions, and single baroreceptor units from atherosclerotic animals demonstrated less sensitivity to non-pulsatile pressure ramps applied to the isolated aortic arch. Li and colleagues (1966) recorded carotid baroreceptor activity from the whole carotid sinus nerve in rabbits after 7-8 months on a high cholesterol diet, and found a reduction in the increase in activity in response to ramps of non-pulsatile pressure relative to responses from untreated animals [16]. Despite the presence of atherosclerotic lesions, carotid bifurcation distensibility in treated animals was not significantly different from that of controls, which ruled out a reduction in strain as the cause of lower baroreceptor activity. Acute exposure of the carotid bifurcation to catalase increased maximum baroreceptor activity of treated animals by 25%, with no effect on controls. They concluded that reactive oxygen species produced by atherosclerotic lesions may have reduced the activity of carotid baroreceptors in atherosclerotic animals. While these two studies both found that atherosclerosis affected arterial baroreceptors, they proposed quite different mechanisms for its effect: one mechanical, the other chemical. In these studies, baroreceptor responses were examined under unnatural conditions: baroreceptor regions were isolated from the general circulation, and they were exposed to physiological saline solution with non-pulsatile pressure ramps applied. Any specific effects of pulsatile pressure and flow or blood-borne substances on the arterial baroreceptors could not have occurred.
As far as we are aware, no one has examined the effects of atherosclerosis on both arterial baroreceptors and the baroreflex control of arterial pressure, HR, and SNA, in the same animal or animal model. In the study reported here, we addressed several outstanding questions in young male New Zealand White rabbits fed an atherogenic diet for 14 weeks. We started with the hypothesis that the development of atherosclerosis would result in lesions in the aortic arch, reductions in aortic arch distensibility, aortic baroreceptor activity, and baroreflex control of HR and RSNA. The questions we asked were: (i) How does the development of atherosclerosis affect responses of aortic baroreceptors in situ exposed to natural pulsatile blood flow and pressure? (2) If changes in aortic baroreceptor behaviour do occur, do they reflect the extent of atherosclerotic lesions and/or changes in aortic distensibility, and are they accompanied by alterations in baroreflex control of HR and RSNA?
The working hypothesis for this study was that treatment will produce atherosclerotic lesions in the larger conduit arteries (including the aortic arch and carotid sinus), an increase in Mean Arterial Pressure (MAP) and Pulse Wave Velocity (PWV), reduce the discharge of arterial baroreceptors, and influence the baroreflex control of HR and RSNA such that the range of baroreflex control is reduced and the response curve is right-shifted.
This study conformed with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH publication No. 85-23, revised 1996), and was approved by the University of Otago Committee on Ethics in the Care and Use of Laboratory Animals.
From one week after weaning, 7-week-old male New Zealand White rabbits (1.5–2 kg) were placed on one of two isocaloric diets for 14 weeks: standard rabbit pellet chow (control group; n = 24); a custom rabbit chow containing 0.5% cholesterol, 3% peanut oil, and 3% coconut oil (treated group; n = 24). Water was available ad libitum. Animals were housed individually at a constant temperature (~20°C) and 12-hour light/dark cycle. Blood samples (1 mL) were taken from a central ear artery at the beginning of the feeding program and weeks 7 and 14 for plasma lipid profiles (see Supplemental material online).
Before the study on which this report is based, a pilot study was performed, which looked for the presence of atherosclerosis in 5 groups of animals (4 animals/group): treated animals after 6, 10, and 14 weeks on the atherogenic diet, and control animals at the 6 and 14-week timepoints (see Supplemental material online). Based on the histological data from the pilot study, a 14-week period of dietary treatment was selected.
Blood lipid analysis was performed on animals in a pilot study (12 treated and 8 control animals). Every 2 weeks, blood (1 mL) was taken from a central ear artery, centrifuged for ten minutes at 2000 g and plasma separated. High-Density Lipoprotein (HDL), total cholesterol, and triglyceride concentrations were measured in the plasma using commercial enzymatic kits (Roche/Boehringer, Mannheim, Germany) and automated methods. Low-Density Lipoprotein (LDL) concentration was calculated using the Friedewald equation [23a].
We investigated several components of the arterial baroreflex. In Experiment 1 relationships between multi-fibre (Expt. 1a) or single-fibre aortic baroreceptor activity (Expt. 1b), and aortic pressure were examined in the anaesthetised animal. Aortic distensibility was calculated from data obtained during these experiments. In Experiment 2 we analysed the baroreflex control of HR and RSNA in the conscious (Expt. 2a) and anaesthetised (Expt. 2b) animal. Observations on multifibre preparations of the ADN (as in Expt. 1a) were also made in many animals used in Expts 1a and 2b. In addition to experiments in the living animal, histological examination of the aortic arch and left ADN was performed (Expts 1a, 1b and 2b).
Animals were anaesthetised with α-chloralose (50 mg kg-1) and urethane (1 g kg-1) administered through a marginal ear vein. During the experiment, limb withdrawal and blink responses were monitored and further anaesthetic infused as required (~5% of the initial dose). For measurement of aortic pressure, a 3F catheter-tip manometer (SPR-249, Millar Instruments, Houston, TX, USA) was advanced to the apex of the aortic arch via the right femoral artery. The chest was opened between the fourth and fifth ribs, and end-expiratory pressure was set at 3-5 cmH2O. Perivascular cuffs (5 mm diameter, In Vivo Metric, Healdsburg, CA, USA) were secured around the inferior vena cava and descending thoracic aorta just rostral to the diaphragm. To maintain cardiac filling pressure 10 mL of dextran solution (50 g L-1; MW 100,000–200,000, Sigma-Aldrich Corp. St. Louis, MO, USA) was injected intravenously. Ultrasonic crystals (5 MHz, 2 mm diameter; Triton Technology, San Diego, CA, USA) were fixed to the thoracic aorta 4-5 cm caudal to the apex of the aortic arch; aortic diameter was measured using a sonomicrometer (Model 6, Triton Technology).
A preliminary set of descriptive data was obtained 30 minutes after surgery and basic instrumentation had been completed, but before the chest was opened. First, aortic pressure was recorded for 10 minutes to obtain a value for Mean Arterial Pressure (MAP). Next, velocity of the Pulse Wave (PWV) between the aortic arch and iliac arteries was measured (see Supplemental material online).
Experiment 1a response of multifibre preparations of the left ADN to a pressure ramp
The left ADN was exposed after the neck had been opened widely through an incision from the chin to the top of the sternum. The ADN was cut at its junction with the superior laryngeal nerve, then placed over a pair of platinum electrodes for recording multifibre action potentials (see Supplemental material online).
To examine baroreceptor responses to a full range of arterial pressure, arterial pressure ramps that rose by ~2 mmHg s-1 from ~25 to ~140 mmHg were produced by inflation, then deflation, of the inferior vena cava cuff, followed by inflation of the aortic cuff. Three or more pressure ramps were applied and the data from these were pooled.
Multi-fibre ADN baroreceptor activity was quantified by integration (1-s time constant decay). Baroreceptor response curves relating integrated activity to aortic pressure were fitted with a third-order polynomial equation. Initial inspection of the response curves suggested that, in the treated animals, there was less integrated activity at higher values of MAP. To quantify this, activity was normalised for each curve using the level of activity recorded at the ramp pressure of 90 mmHg as a reference value (100 units). Next, the Area Under the Curve (AUC) between 90 and 140 mmHg was measured.
Preparation for this experiment was the same as for Expt. 1a. To record single-fibre baroreceptor activity, the ADN was split repeatedly into small filaments, until a signal from a single afferent fibre could be clearly discriminated. Responses to pressure ramps were obtained and recorded as described for Expt. 1a.
Frequency of single-fibre baroreceptor activity was obtained using the cyclic variable software in Chart v5.5.6. Single fibre baroreceptor response curves relating action potential frequency to aortic pressure were fitted with a four-parameter logistic curve. To characterise and compare baroreceptor response curves, values for several parameters were extracted (threshold pressure, threshold activity, BP50, BP50 activity, saturation pressure, activity at saturation pressure, and gain).
To record RSNA, an electrode was implanted around the left renal nerve. Animals were sedated with ketamine and medetomidine injected s.c. (Ketalar, 15 mg kg-1; Dormitor, 0.25 mg kg-1). After intubation, animals were maintained on 3% halothane in O2, while respiratory rate and blink reflex were monitored. The electrode cable was brought through a subcutaneous tunnel to the back of the neck where it was retained in a subcutaneous pocket (see Supplemental material online for full details).
Five days of recovery elapsed before Expt. 2a was conducted. For the duration of the experiment, the animal was gently restrained in a box that prevented excessive movement that would interfere with procedures and data collection. Under local anaesthesia (lignocaine hydrochloride, Paraderm Plus, New Zealand), a central ear artery and a marginal ear vein were catheterised (24G and 22G catheters, respectively, BD Insyte-N, Becton Dickinson Pty Ltd., NSW, Australia). Dead space was filled with heparinised saline (2 units mL-1, David Bull Laboratories, New Zealand). Arterial pressure was measured using a pressure transducer (P23ID, Statham, Hato Rey, Puerto Rico). To record RSNA the electrode cable was retrieved from its pocket after local anaesthetic was applied (Lignocaine, 2% Xylocaine). Signals from the renal nerve were amplified and filtered using a differential preamplifier (DAM80), with high and low pass filters set at 100 Hz and 10 kHz. RSNA was sampled, digitised and recorded using the same parameters described for ADN activity.
Arterial pressure was recorded for 10 minutes to obtain conscious MAP. Next, baroreflex responses of HR and RSNA were obtained. A slow infusion of sodium nitroprusside (1 mg mL-1) reduced MAP to ~40 mmHg, then a slow infusion of phenylephrine (1 mg mL-1) raised MAP to 140 mmHg at a rate of 2 to 5 mmHg s-1. This procedure (SNP+PE) was performed three times, with a 10-min interval between tests.
HR was calculated from the arterial pressure signal using cyclic variable software in Chart v5.5.6.
Expt. 2b was conducted 1-2 days after Expt. 2a. The animal was anaesthetised, and the surgical and technical preparations described for Expt. 1a were completed. Electrode terminals from the renal nerve preparation were retrieved and set up for recording of RNSA as described for Expt. 2a.
Following instrumentation, aortic pressure and renal sympathetic nerve activity were recorded for 10 minutes to establish baseline aortic pressure, heart rate, and RSNA values. Two methods were used to examine baroreflex control of RSNA in the anaesthetised animal. In the first, baroreflex control of RSNA was determined from responses to infusion of sodium nitroprusside and phenylephrine as described previously. To investigate the contribution of the aortic baroreceptors alone to baroreflex control of RSNA, the drug procedure was performed with both common carotid arteries temporarily occluded (BCCA) which effectively eliminated responses from the carotid sinus baroreceptors.
In the second method, perivascular cuffs were placed around the inferior vena cava and descending thoracic aorta (described in detail for Expt. 1). Measurements of baroreflex control of RSNA in response to ramp changes in arterial pressure were obtained both before and during BCCA.
Data were analysed after fitting with a four-parameter logistic curve (see Expt. 2a).
Histology was performed on the aortic arch (n = 24 from each group). At the end of Expts. 1a, 2b, and 2b, the animal received 2 mL heparinised saline (1000 units mL-1 intravenously) and euthanized 5 min later with an overdose of α–chloralose and urethane. A catheter was inserted just distal to the aortic arch for retrograde perfusion of fluids to achieve tissue fixation (see Supplemental material online), with fluid drained by a catheter in the right atrium.
The aortic arch was embedded, orientated longitudinally in paraffin, and serial sections (4-µm thick) were obtained at the point of maximum luminal diameter, processed and examined (see Supplemental material online). The thickness of the intima along the inner curvature of the aortic arch - from the ascending aorta to the ligamentum arteriosum - was quantified28 using calibrated image analysis software (ImageJ, ImageJ.org).
The left ADN was retrieved from 9 control and 9 treated animals at the completion of Experiment 2b. Before removing the ADN, the anaesthetised animal was administered 2 mL heparinized 0.9% saline i.v. (1000 units mL-1). Five minutes later, the animal was euthanized with intravenous chloralose and urethane. Fixation was carried out as described above (section 2.09).
The ADN was cut close to its junction with the superior laryngeal nerve and separated from surrounding tissue, and a portion prepared for histology (see Supplemental material online). A ~2-cm length of the ADN was removed and immersion fixed in 2% glutaraldehyde in 0.1 M cacodylate buffer (3 x 10 minute washes in 0.1 M cacodylate buffer), followed by 2 x 10-minute washes in 0.1 M cacodylate buffer. The nerve was refrigerated in 0.1 M cacodylate buffer until dehydration and infiltration (see Supplementary material online).
ADN sections were viewed using a Phillips CM100 transmission electron microscope (Phillips/FEI Corporation, Eindhoven, Holland), and images were photographed with a MegaView III digital camera (Olympus Soft Imaging Solutions GmbH, Munster, Germany). Images were taken at 4200x magnification to ensure that each fibre type could be clearly delineated. Adobe Photoshop CS was used to construct a photomontage from the multiple images.
Fibre populations were counted using Counter 4 (Paz Software, details). Each photomontage was counted at least twice to confirm reliability of the counting procedure. For 100 randomly selected A- and C-fibres, fibre diameter was recorded as the mean of the longest and shortest axes. Histograms of the size distribution of the myelinated (A-fibres) and unmyelinated (C-fibres) fibres were constructed by pooling axons, by mean diameter, into bins at intervals of 0.5-µm and 0.1-µm, respectively.
Diameter and distensibility of the descending thoracic aorta were obtained from data collected during pressure ramps in Experiment 2.6. Measurements of diameter and distensibility were made at diastolic aortic pressures of 40, 80, 90, and 140 mmHg (see Supplemental material online and Supplemental Figure SC). Subsequently, diameter and distensibility data were normalised using the diameter and distensibility of the aorta recorded at a diastolic pressure of 40 mmHg (reference pressure). At 40 mmHg, the aortic wall should be at, or close to its unstressed state.
We also examined values for diameter and distensibility after they were normalised to take into account: first, variability in aortic diameter as a consequence of variability in body mass, and, secondly, the deviation of a measurement from the true diameter. Any deviation from an exact placement of ultrasonic crystals would report the length of a chord that was close to, but slightly shorter than the true diameter. Subsequently, diameter and distensibility data were normalised using the diameter and distensibility of the aorta recorded at a diastolic pressure of 40 mmHg (reference pressure). At 40 mmHg the aortic wall should be at, or close to, its unstressed state.
In all figures, data are presented as group means ± 1 standard deviation. In the text, mean values are shown with the [90% confidence interval].
Between-group differences were examined using Student’s t-test. For comparisons where an a priori hypothesis had been made, a single-tailed t-test was applied. In baroreflex experiments carried out in anaesthetised animals, repeated measures analysis (paired t-test) was used on data collected before and after common carotid artery occlusion [24-28].
For 21-week-old animals, body mass of the treated group (3.58 [3.52, 3.63] kg, N = 24) was similar to values for the control group (3.64 [3.57, 3.71] kg, N = 24]); p = 0.58).
There were extensive atherosclerotic lesions along the inner curvature of the aortic arch of all treated animals (see Supplemental material online, Figure R1), while the intima of control animals remained normal. Xanthoma lesions, present in all treated animals, ranged from small focal foam cell accumulations–forming fatty streaks–to large advanced lesions that contained abundant fibrous extracellular material, covering a substantial portion of the aortic arch. Lesions varied in both extent and thickness (see Supplemental material online, Figure R2), covering 65 ± 25% (mean ± SD) of standardised length (range, 11–100%). Mean intimal thickness was 46 ± 34% (range, 4–123%). Lesions were also observed in the outer curvature of the arch (location of aortic baroreceptors with afferent fibres running in the left ADN) and the origin of the brachiocephalic artery (location of baroreceptor fibres with afferents running in the right ADN) [29]. There were no atherosclerotic lesions in the carotid sinus of treated animals.
Under anaesthesia, MAP was higher in the treated group compared with the control (99.9 [97.2, 102.7] versus 95.6 [93.5, 97.7], respectively, N = 24 for each group; p = 0.050). Pulse wave velocity was about 10% higher in the treated group (5.64 [5.54, 5.74] versus 5.07 [4.98, 5.16] m/s in control,
N = 24 for each group; p < 0.0001). In the treated animals, PWV correlated with the intimal thickness of the aortic arch (see Supplemental material online, Figure R3).
The atherogenic had no overt effects on the health and well-being of animals, and no signs of anorexia or weight loss attributable to cholesterol toxicity.
In treated animals, total plasma cholesterol, LDL, and triglycerides increased over the first 6 weeks of the study, then levelled off. In 12 treated and 8 control animals, total cholesterol, LDL, and triglyceride concentrations were, respectively: 34.2 [28.9,39.4] vs. 1.5 [1.3,1.7] mmol L-1, p < 0.0001; 32.6 [27.4,37.8] vs. 1.0 [0.5,1.5] mmol L-1, p < 0.0001; and 1.9 [1.6,2.2] vs. 0.5 [0.3,0.8] mmol L-1, p < 0.0001. Plasma HDL values remained stable throughout treatment: 0.83 [0.6,0.9] vs. 0.77 [] mmol L-1, in treated and control animals, respectively, p = 0.72. The degree of hypercholesterolemia seen in this study was similar to that seen in previous studies utilising a diet-induced hypercholesterolemic rabbit model of atherosclerosis [29a].
Multifibre baroreceptor recordings: Multifibre baroreceptor response curves recorded from the left CSN of treated and control animals were similar (Figure 3.1A). At 140 mmHg, normalised integrated activity was 137 [129,144] and 141 [133,149], in 10 treated and 10 control animals, respectively, p = 0.29.
![Integrated baroreceptor activity during a pressure ramp. Lines show the normalised mean activity from treated (dashed) and control (solid) animals. Representative error bars show 1SD.
(A.) Mean of normalised integrated CSN activity from 10 treated and 10 control animals. (B) Mean of normalised integrated ADN activity from 22 treated and 20 control animals. The mean area under the curve at pressures above 90 mmHg was smaller in treated than in control animals (4650 [4174, 4988] versus 5570 [5220, 5919] au; p < 0.001, single-tailed t-test.](https://www.igminresearch.com/articles/figures/igmin340/igmin340.g001.png) Figure 3.1: Integrated baroreceptor activity during a pressure ramp. Lines show the normalised mean activity from treated (dashed) and control (solid) animals. Representative error bars show 1SD. (A.) Mean of normalised integrated CSN activity from 10 treated and 10 control animals. (B) Mean of normalised integrated ADN activity from 22 treated and 20 control animals. The mean area under the curve at pressures above 90 mmHg was smaller in treated than in control animals (4650 [4174, 4988] versus 5570 [5220, 5919] au; p < 0.001, single-tailed t-test.
Figure 3.1: Integrated baroreceptor activity during a pressure ramp. Lines show the normalised mean activity from treated (dashed) and control (solid) animals. Representative error bars show 1SD. (A.) Mean of normalised integrated CSN activity from 10 treated and 10 control animals. (B) Mean of normalised integrated ADN activity from 22 treated and 20 control animals. The mean area under the curve at pressures above 90 mmHg was smaller in treated than in control animals (4650 [4174, 4988] versus 5570 [5220, 5919] au; p < 0.001, single-tailed t-test.In recordings from the ADN, at pressures above 90 mmHg, the curve for mean activity in treated preparations diverged from the control curve (Figure 3.1B). At 140 mmHg, normalised integrated activity was 183 [173, 194] and 201 [192, 210], in 22 treated and 20 control animals, respectively, p = 0.027. The mean Area Under the Curve (AUC) was smaller in treated than in control animals (4650 [4174, 4988] versus 5570 [5220, 5919] au; N = 22 and 20, respectively, p < 0.001). In treated animals, the size of the AUC correlated negatively with intimal thickness (see Supplemental material online, Figure R4).
The means of responses from single baroreceptor fibres of the left ADN are shown in Figure 3.2. Single fibre activity from treated and control animals was similar over the full pressure ramp and reached similar maximum firing rates at 140 mmHg (76.2 [72.0, 80.5] and 73.9 [70.8, 77.0] Hz; N = 52 and 73, respectively, p = 0.98). Based on the maximum firing frequency of these recordings, we concluded that they were taken from myelinated A baroreceptor fibres [30].
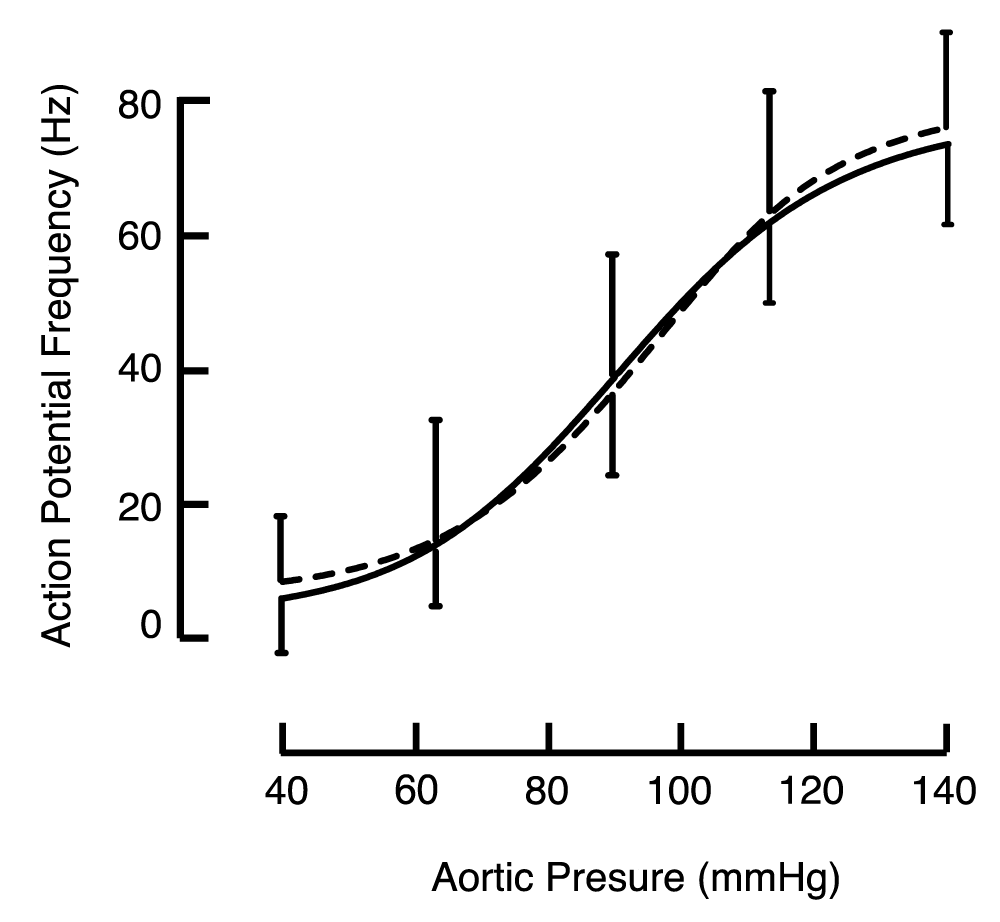 Figure 3.2: Frequency of action potentials in single baroreceptor fibres from the ADN during a pressure ramp. Data from treated (dashed) and control (solid) animals. Representative error bars show 1SD.
Figure 3.2: Frequency of action potentials in single baroreceptor fibres from the ADN during a pressure ramp. Data from treated (dashed) and control (solid) animals. Representative error bars show 1SD.Since there were no atherosclerotic changes to the carotid bifurcation in treated animals, and because the multifibre baroreceptor responses from carotid sinus baroreceptors were similar in treated and control groups, we chose not to obtain single fibre recordings from the CSN.
The four-parameter logistic curve fit the HR and RSNA data well; R2 exceeded 0.99 over the full pressure range for all individual curves.
At rest, before reflex testing, MAP was substantially higher in the treated group (80.1 [77.3,82.9] versus 70.9 [69.9, 71.9]; N = 7 and 9, respectively, p < 0.001), while HR and RSNA were similar (see Supplemental material online, Table R1). The baroreflex curve for RSNA, but not for HR, was right-shifted in treated animals (Figure 3.3 and Supplemental Table R1).
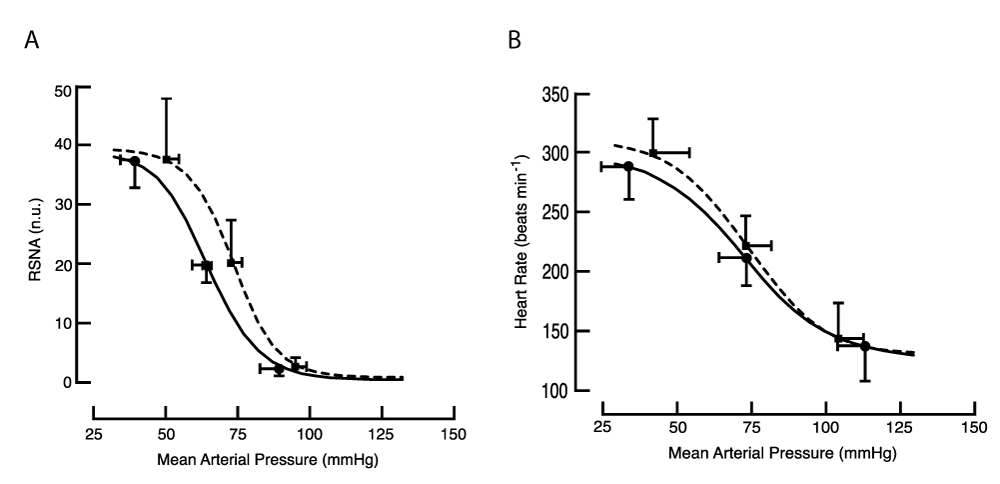 Figure 3.3: Baroreflex curves in the conscious animal. Symbols identify the location of mean values for threshold, mid-point, and saturation of the baroreflex. (A) RSNA: the mean of fitted curves obtained from 5 treated (dashed line) and 8 control animals (solid line). The baroreflex curve for RSNA was right-shifted in treated animals; at threshold p = 0.01, at mid-point p = 0.02, single-tailed t-test. (B) HR: the mean of fitted curves obtained from 7 treated and 8 control animals.
Figure 3.3: Baroreflex curves in the conscious animal. Symbols identify the location of mean values for threshold, mid-point, and saturation of the baroreflex. (A) RSNA: the mean of fitted curves obtained from 5 treated (dashed line) and 8 control animals (solid line). The baroreflex curve for RSNA was right-shifted in treated animals; at threshold p = 0.01, at mid-point p = 0.02, single-tailed t-test. (B) HR: the mean of fitted curves obtained from 7 treated and 8 control animals.The reflex response of RSNA was examined using both drug and cuff methods. Before examining the baroreflex, resting MAP and RSNA were similar in treated and control animals (see Supplemental material online, Table R2).
Baroreflex responses obtained with the drug method (Figure 3.4 A and B, Supplemental Table R2) were similar for the treated and control groups. During BCCO, the curves for both treated and control animals were right-shifted (Figure 3.4 and Supplemental Table R2).
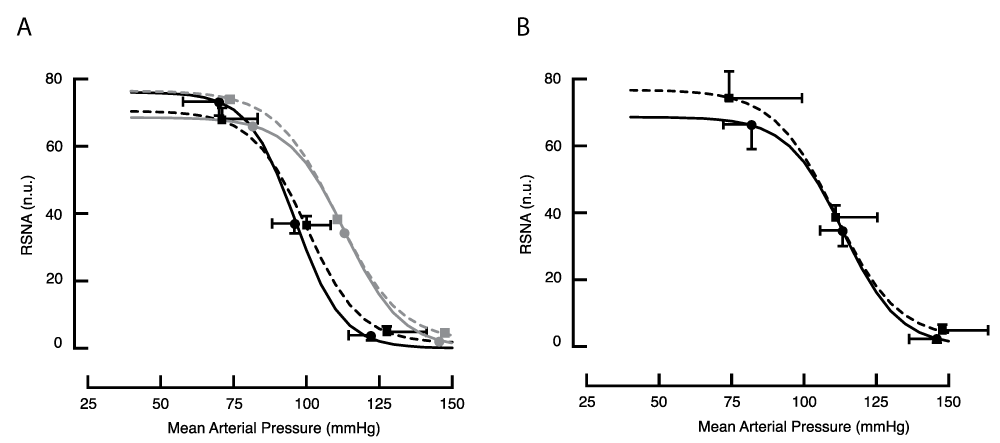 Figure 3.4: Baroreflex response curves for RSNA in the anaesthetised animal obtained with the drug method, (A) before and (B) during BCCO. Lines drawn through the means of fitted curves from 5 treated (dashed line) and 8 control animals (solid line). To illustrate the effect of BCCO on these curves, the lighter grey lines in panel A reproduce the lines drawn in panel B.
Figure 3.4: Baroreflex response curves for RSNA in the anaesthetised animal obtained with the drug method, (A) before and (B) during BCCO. Lines drawn through the means of fitted curves from 5 treated (dashed line) and 8 control animals (solid line). To illustrate the effect of BCCO on these curves, the lighter grey lines in panel A reproduce the lines drawn in panel B.Using the cuff method, the value of RSNA at threshold was about 75% the value seen in the drug method (Figure 3.5 A and B, and Supplemental Table R3). This may be a result of opening the chest to place vascular occlusion cuffs and the consequent removal of negative intrapleural pressure. Nevertheless, the form of the response curves obtained with the cuff method resembled those obtained using the drug procedure. Arterial pressure at threshold, mid-point, and saturation before BCCO were similar in treated and control animals. During BCCO, both curves were right-shifted (Table R3).
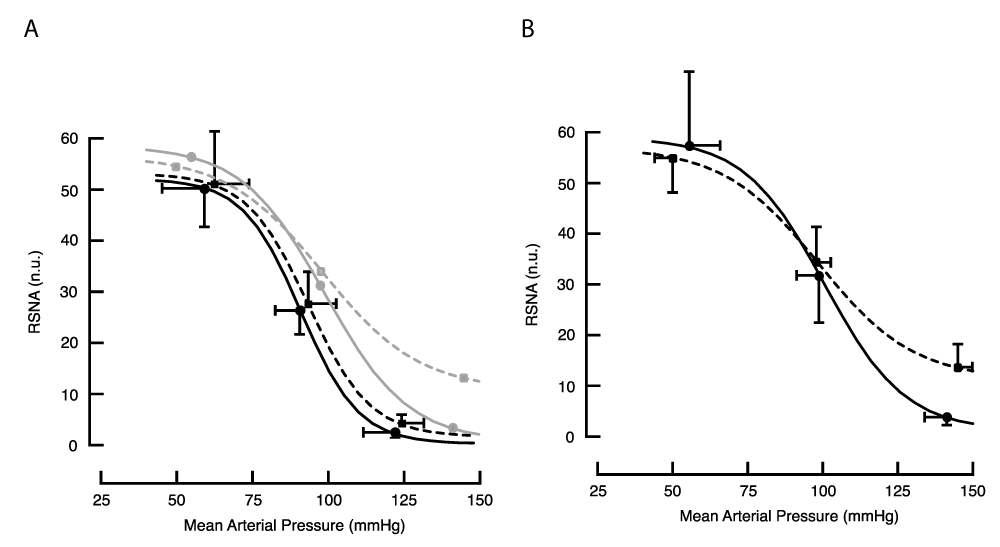 Figure 3.5: Baroreflex response curves for RSNA in the anaesthetised animal obtained with the cuff method, (A) before and (B) during BCCO. Lines drawn through the means of fitted curves from 5 treated (dashed line) and 7 control animals (solid line). To illustrate the effect of BCCO on these curves, the lighter grey lines in panel A reproduce the lines drawn in panel B. During BCCO, RSNA at saturation was greater in treated animals; p = 0.01, single-tailed t-test.
Figure 3.5: Baroreflex response curves for RSNA in the anaesthetised animal obtained with the cuff method, (A) before and (B) during BCCO. Lines drawn through the means of fitted curves from 5 treated (dashed line) and 7 control animals (solid line). To illustrate the effect of BCCO on these curves, the lighter grey lines in panel A reproduce the lines drawn in panel B. During BCCO, RSNA at saturation was greater in treated animals; p = 0.01, single-tailed t-test.An interesting feature of response curves obtained by the cuff method during BCCO was a weaker restraint of RSNA at the higher pressures in treated animals. RSNA at saturation was 12.5 [8.4, 16.6] and 3.9 [2.6, 5.2] n.u. in 5 treated and 7 control animals, respectively, p = 0.01. This contrasts with the complete inhibition of RSNA at higher pressures in all reflex tests conducted in this study, including tests on control animals during BCCO.
The dimensions of the left ADN and a description of the populations of A- and C-fibre afferent fibres from 9 treated and 9 control animals are shown in Table 3.1. The values for all 6 variables were similar in treated and control animals. The populations of both A- and C-fibres had a unimodal size distribution (see Supplemental material online, Figure R5).
A-fibres had similar diameters in treated and control animals (2.44 [2.36, 2.52] and 2.43 [2.36, 2.50] µm; N = 900 from each group; p = 0.94). Likewise, C fibre diameter was similar in treated and control animals (0.63 [0.60, 0.66] and 0.66 [0.63, 0.69] µm, respectively; N = 900 from each group; p = 0.18).
For reasons stated in Methods, we examined these data in both absolute and normalized units using the diameter at 40 mmHg (See Supplemental material online, Figure R6 and Table R4). The mean diameter of the thoracic aorta at 40 mmHg was similar in treated animals and control animals. Normalised diameters at 80, 100, and 140 mmHg were smaller in treated versus control animals. Aortic compliance was lower at 80 and 100 mmHg, but not at 140 mmHg, in treated versus control animals.
Young male rabbits placed on an atherogenic diet for 14 weeks developed substantial atherosclerotic lesions in the aortic arch, and lipid deposits appeared at all aortic flow dividers. Relative to measurements in control animals, compliance of the thoracic aorta 5 cm below the arch was lower in treated animals, while PWV along the aorta was higher. Furthermore, PWV was uniform along the full length of the aorta, suggesting that aortic wall compliance was lower throughout its length, not just at segments that exhibited gross atherosclerotic lesions.
The extent of atherosclerotic lesions in the aortic arch showed modest associations with PWV. However, these lesions, together with lipid deposits at aortic flow dividers, cannot entirely account for the higher PWV. It appears that the dietary treatment either directly, or secondarily in response to these gross atherosclerotic changes, resulted in arteriosclerosis along the full length of the aorta [31]. Whether the variability of PWV in treated animals depended directly on the extent of the atherosclerotic lesions or reflected a more general variability in the animals’ responses to dietary treatment cannot be determined from our data. Inflammation may have been the common driver for the coincident development of atherosclerotic and arteriosclerotic changes in the treated animals [32,33].
In the conscious animal prepared for baroreflex study, mean control values for MAP (71 mmHg) were similar to those reported in other studies [34,35], while MAP in the treated group was ~10% higher. In the larger group of animals studied under chloralose-urethane anaesthesia, the mean value for MAP was 96 mmHg for the control group, similar to that reported by Dorward, et al. [24], and was ~4% higher in the treated group. Arterial hypertension generated by atherosclerosis may be attributed to reduced renal function [36,37]. This could be a result of structural changes - a narrowing of renal arteries that requires a higher driving pressure [11], or an increase in RSNA [38,39]. We did not examine the renal vasculature, but it seems unlikely that a substantial narrowing of renal arteries would have occurred over the 14-week treatment period. On the other hand, RSNA may have been increased in response to changes in the arterial baroreflex or in central generations of peripheral sympathetic activity [22]. In the current study, treatment shifted the mid-point of the baroreflex curve for RSNA to a higher arterial pressure.
Our observations on ADN baroreceptor activity suggest a mechanism for elevated MAP and altered baroreflex control in treated animals. At MAPs above 100 mmHg, total integrated multifibre baroceptor activity (AUC) was lower in treated animals, and we attributed this to a substantial reduction in C-fibre activity in treated animals. While pressure-activity curves recorded from single myelinated baroreceptor afferents (presumptive A-fibre baroreceptor afferents) were similar in treated and control animals over the full range of MAP examined (40-100 mmHg), C-fibre baroreceptors have a higher pressure threshold (70-140 mmHg versus 35-90 mmHg) and will contribute much less to integrated activity recorded at a MAP lower than 100mmHg [40].
In treated animals, integrated ADN activity at a MAP of 140 mmHg was ~10% less than control values. This represents a large effect of treatment on C-fibre activity since C-fibre activity contributes considerably less than A-fibre activity to total integrated activity. First, extracellular action potentials produced by C-fibres are smaller than by A-fibres, and will make a smaller contribution to the total integrated signal40. Secondly, maximum firing rates for C-fibres are ~15% of those for A-fibres [41]. The number and size of C-fibre afferents in the ADN were not affected by the treatment, so it’s unlikely that conduction of action potentials generated by the C-fibre sensory terminals in treated animals was compromised.
A lower C fibre activity in treated animals could result from reduced mechanical stimulation or from inflammation of the vessel wall. Sensory endings of C fibres are located in the media of the aortic wall [28,42], and are thought to be activated by compression rather than by circumferential strain [30]. Consistent with this, the AUC was negatively associated with the extent of atherosclerotic lesions in the arch - the thicker the aortic wall, the lower the baroreceptor C-fibre discharge. Reactive oxygen species produced by atherosclerotic lesions [43,44] may tonically suppress baroreceptor discharge [16]. Again, AUC may be expected to correlate negatively with the severity of atherosclerotic lesions.
Altered baroreflex regulation of RSNA in treated animals is consistent with the lower ADN baroreceptor activity recorded at higher arterial pressures; baroreflex curves for RSNA were right-shifted. When BCCO was applied to minimise input from the carotid sinus baroceptors, high pressure was unable to fully silence RSNA in treated animals, indicating that at very high pressures, aortic baroreceptor input was now insufficient to fully inhibit RSNA. In the rabbit, a normally functioning baroreflex from the carotid sinus baroreceptors may compensate completely for any deficit in control by aortic baroreceptors [45]. Nervous input to the CNS from the four primary arterial baroreceptor areas operates in an occlusive fashion to determine the baroreflex response, and elimination of carotid sinus baroreceptor input (which appeared to be unaffected by treatment) unmasked the aortic baroreceptor deficit [46]. Since we found that baroreceptor A-fibre activity at high MAP was unaltered by treatment, a lower baroreceptor C-fibre activity at high MAP in treated animals appears the most likely cause of incomplete reflex inhibition of RSNA at high MAP. While an effect of the treatment on central integration of the reflex cannot be ruled out, it seems less likely since, in the absence of BCCO, high MAP fully suppressed RSNA in treated animals.
While the atherosclerotic diet always produced lesions in the aorta, the extent of these lesions showed considerable variation between treated animals. Substantial associations between the extent of the lesions and aortic PWV and aortic C-fibre activity were observed. In our interpretations of these associations, we have treated the aortic lesions as the independent variable. Thus, while the experimental model has a single independent variable applied at a single level, in practice, it can produce variable degrees of atherosclerotic change, which may be useful in determining causation.
The time-frame over which atherosclerotic changes were developed in the rabbit model is much shorter than the progression of atherosclerosis in humans. While results from this study indicate that atherosclerotic lesions in the aortic arch are associated with altered aortic baroreceptor activity and reflex control of RSNA, it is not possible to state that atherosclerosis in humans would be accompanied by similar changes in the arterial baroreceptor mechanism.
The effects of manipulations used to examine the arterial baroreflex are not limited to alterations in MAP to which the arterial baroreceptors are subjected, and which is subsequently treated as the independent variable. First, the cardiovascular effects of both the drug and cuff methods also affect the level of stimulation of other vascular and cardiac receptors that can influence RSNA, and the two different methods differ in these effects [27]. Secondly, the drug method can cause pressure-independent changes in baroreceptor activity [47]. SNP can relax, and PE can contract smooth muscle in the media, modifying the mechanical strain sensed by A-fibre baroreceptors. Meanwhile, changes in smooth muscle tension may alter compressive forces that activate C-fibre baroreceptors. There is also the possibility that PE can directly activate C-fibre baroreceptors. Some differences in the reflex curves obtained during BCCO from treated animals using the drug and cuff methods may reflect the influence of these confounding factors; the lack of full suppression of activity at high MAP was strong only for the cuff method; the value of RSNA at threshold with the cuff method was about 75% the value seen in the drug method.
Fourteen weeks on a diet that produced atherosclerotic changes to the walls of the rabbit aorta were sufficient to cause modest hypertension, modify both the activity from and the reflex responses produced by the aortic baroreceptors. This was attributed to a reduction in the responsiveness of the unmyelinated C-fibre baroreceptors.
It is well established that hypertension, even at modest levels, acts as one of the primary drivers for atherosclerosis. Now we have found that atherosclerosis interferes with unmyelinated baroreceptor activity which, reflexively, may raise arterial pressure. Thus there may be a vicious cycle, with bidirectional causality between hypertension and atherosclerosis. The literature includes data suggesting that even modest values for arterial pressure (starting at or below 120/80) should be considered as hypertensive, [48], and may be associated with atherosclerosis. This stresses the need for identification and treatment of atherosclerosis and hypertension early in life.
The authors are grateful to Dr Gregory T Jones for preparing material for histological analysis and to Dave Matthews for assistance during surgery for chronic implantation of renal sympathetic nerve electrodes.
This study was supported by funds from the Department of Physiology, University of Otago, the Otago Medical Research Foundation, and the National Heart Foundation of New Zealand.
The data that support the findings of this study are available upon request from the corresponding author.
1. Incalcaterra E, Accardi G, Balistreri CR, Caimi, G, Candore G, Caruso M, et al. Pro-Inflammatory Genetic Markers of Atherosclerosis. Curr Atheroscler Rep. 2013; 15:329–337. doi: 10.1007/s11883-013-0329-5. PMID: 235916722. Jinnouchi H, Sato Y, Sakamoto A, Cornelissen A, Mori M, Kawakami R, et al. Calcium deposition within coronary atherosclerotic lesions: Implications for plaque stability. Atherosclerosis 2020; 306:85-95. doi: 10.1016/j.atherosclerosis.2020.05.017. PMID: 32654790.3. Poznyak AV, Sadykhov NK, Kartuesov AG, Borisov EE, Melnichenko AA, Grechko AV, et al. Hypertension as a risk factor for atherosclerosis: Cardiovascular risk assessment. Front Cardiovasc Med 2022; 9:959285. doi.org/10.3389/fcvm.2022.959285. PMID: 36072873.4. Libby P. The changing landscape of atherosclerosis. Nature 2021; 592:524–533. doi.org/10.1038/s41586-021-03392-8. PMID: 33883728.5. Lehmann N, Erbel R, Mahabadi AA, Kälsch H, Möhlenkamp S, Moebus S, et al. Accelerated progression of coronary artery calcification in hypertension, but also prehypertension. J Hypertens 2016; 34:2233–2242. doi: 10.1097/HJH.0000000000001080. PMID: 27512973.6. Gonzalez-Guerra A, Roche-Molina M, García-Quintáns N, Sánchez-Ramos C, Martín-Pérez D, Lytvyn M, et al. Sustained Elevated Blood Pressure Accelerates Atherosclerosis Development in a Preclinical Model of Disease. Int J Mol Sci 2021;22:8448. doi: 10.3390/ijms22168448. PMID: 34445154.7. Al-Mashhadi AL, Nasr ZP, Mortensen MB, Lewis EA, Camafeita E, Ravlo K, et al. Local Pressure Drives Low-Density Lipoprotein Accumulation and Coronary Atherosclerosis in Hypertensive Minipigs. Am Coll Cardiol 2021;77:575–589. doi: 10.1016/j.jacc.2020.11.059. PMID: 33538256.8. Mautner SL, Mautner GC, Hunsberger SA, Roberts WC. Comparison of composition of atherosclerotic plaques in saphenous veins used as aortocoronary bypass conduits with plaques in native coronary arteries in the same men. Am J Cardiol 1992;70:1380–1387. doi: 10.1016/0002-9149(92)90285-7. PMID: 1442604.9. Moore GW, Smith RR, Hutchins GM. Pulmonary artery atherosclerosis: correlation with systemic atherosclerosis and hypertensive pulmonary vascular disease. Arch Pathol Lab Med 1982;106:378–380. PMID: 6213213.10. Furukawa K, Kawano H, Futakuchi M, Ishijima M, Yamgataga Y, Ueno Y, et al. Pulmonary atherosclerosis in a patient with chronic thromboembolic pulmonary hypertension. Pathol Int 2021;71:164–166. doi:10.1111/pin. 13051. PMID: 33316138.11. Prince M, Tafur JD, White CJ. When and how should we revascularize patients with atherosclerotic renal artery stenosis? JACC Cardiovasc Interv 2019;25:505–517. doi: 10.1016/j.jcin.2018.10.023. PMID: 30898248.12. Cowley AW Jr. Long-term control of arterial blood pressure. Physiol Rev 1992;72:231–300. doi: 10.1152/physrev. 1992.72.1.231. PMID: 1731371.13. Seagard JL, Hopp FA, Drummond HA, Van Wynsberghe DM. Selective contribution of two types of carotid sinus baroreceptors to the control of blood pressure. Circ Res 1993;72:1011–1022. doi: 10.1161/01.res.72.5.1011. PMID: 8477517.14. Thrasher TN. Effects of chronic baroreceptor unloading on blood pressure in the dog. Am J Physiol Regul Integr Comp Physiol 2005:288:R863–R871. doi: 10.1152/ajpregu.00489. PMID: 15563576.15. Lohmeier TE, Barrett AM, Irwin ED . Prolonged activation of the baroreflex: a viable approach for the treatment of hypertension? Curr Hypertens Rep 2005;7:193–198. doi: 10.1007/s11906-005-0009-0. PMID: 15913493.16. Li Z, Mao HZ, Abboud FM, Chapleau MW. Oxygen-derived free radicals contribute to baroreceptor dysfunction in atherosclerotic rabbits. Circ Res 1996;79:802–811. doi: 10.1161/01. res.79.4.802. PMID: 8831504.17. Gianaros PJ, Jennings JR, Olafsson GB, Steptoe A, Sutton-Tyrrell K, Muldoon MF, et al. Greater intima-media thickness in the carotid bulb is associated with reduced baroreflex sensitivity. Am J Hypertens 2002;15:486–491. doi: 10.1016/s0895-7061(02)02923-0. PMID: 12074348.18. Tsekouras NS, Katsargyris A, Skrapar I, Bastounis EE, Georgopoulos S, Klonar C, et al. The role of carotid plaque echogenicity in baroreflex sensitivity. J Vasc Surg 2011;54:93–99. doi: 10.1016/j.jvs.2010.11.121. PMID: 21458208.19. Cox RH, Bagshaw RJ, Detweiler DK. Alterations in carotid sinus reflex control of arterial hemodynamics associated with experimental hyperlipemia in the racing greyhound. Circ Res 1980;46:237–244. doi: 10.1161/01.res.46.2.237. PMID: 7351041.20. Morita H, Nishida Y, Motochigawa H, Watanabe Y, Hosomi H. Depressed baroreflex control of renal sympathetic nerve activity in conscious WHHL rabbits. Cardiovasc Res 1988;22:678–685. doi: 10.1093/cvr/22.10.679. PMID: 2476214.21. Katsuda S, Hosomi H, Shiomi M, Watanabe Y. Impaired baroreflex control of arterial pressure in WHHL rabbits. J Vet Med Sci 1992; 54:983–987. doi: 10.1292/jvms.54.983. PMID: 1420582.22. McCully BH, Brooks VL, Andresen MC. Diet-induced obesity severely impairs myelinated aortic baroreceptor reflex responses. Am J Physiol Heart Circ Physiol 2012;302:H2083–H2091. doi: 10.1152/ajpheart.01200.2011. PMID: 22408022.23. Angell-James JE. Arterial baroreceptor activity in rabbits with experimental atherosclerosis. Circ Res 1974;40:27–39. doi: 10.1161/01.res.40.4.27. PMID: 4809347.23a. Friedewald WT, Levy RI, Fredrickson S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972;18:499–502. PMID: 4337382.24. Dorward PK, Burke SL, Jänig W, Cassell J. Reflex responses to baroreceptor, chemoreceptor and nociceptor inputs in single renal sympathetic neurones in the rabbit and the effects of anaesthesia on them. J Autonom Nerv Syst 1987;18:39–54. doi: 10.1016/0165-1838(87)90133–0. PMID: 3819315.25. Dorward PK, Riedel W, Burke SL, Gipps J, Korner PI. The renal sympathetic baroreflex in the rabbit. Arterial and cardiac baroreceptor influences, resetting, and the effect of anesthesia. Circ Res 1985;57:618–633. doi: 10.1161/01.res.57.4.618. PMID: 4042286.26. Burke SL, Head, GA. Method for in vivo calibration of renal sympathetic nerve activity in rabbits. J Neurosci Methods 2003;127:63–74. doi: 10.1016/s0165-0270(03)00121-3. PMID: 12865149.27. McDowall LM, Dampney RA. Calculation of threshold and saturation points of sigmoidal baroreflex function curves. Am J Physiol Heart Circ Physiol 2006;291;H2003–2007. doi: 10.1152/ajpheart.00219.2006. PMID: 16714364.28. Jones GT, Jiang F, McCormick SP, Dusting GJ. Elastic Lamina Defects Are an Early Feature of Aortic Lesions in the Apolipoprotein E Knockout Mouse. J Vasc Res 2005;42:237–246 doi: 10.1159/000085553. PMID: 15860923.29. Aumonier FJ. Histological observations on the distribution of baroreceptors in the carotid and aortic regions of the rabbit, cat and dog. Acta Anat 1972;82:1–16. doi: 10.1159/000143797. PMID: 4669378.29a. Fan J, Shimoyamada H, Sun H, Marcovina S, Honda K, and Watanabe T. Transgenic rabbits expressing human apolipoprotein(a) develop more extensive atherosclerotic lesions in response to a cholesterol-rich diet. Arterioscler Thromb Vasc Biol 2001;21:88-94. doi: 10.1161/01.atv.21.1.88. PMID: 11145938.30. Thoren P, Munch PA, Brown AM. Mechanisms for activation of aortic baroreceptor C-fibres in rabbits and rats. Acta Physiol Scand 1999;166:167–174. doi:10.1046/J.1365-201X.1999.00556.X. PMID: 10468652.31. Giannattasio C, Failla M, Emanuelli G, Grappiolo A, Boffi L, Corsi D, et al. Local effects of atherosclerotic plaque on arterial distensibility. Hypertension 2001;38:1177–1180. doi: 10.1161/hy1101.095994. PMID: 11711518.32. Makita S, Nakamura S, Hiramori, K. The association of C-reactive protein levels with carotid intima-media complex thickness and plaque formation in the general population. Stroke 2005;36:2138–2142. doi: 10.1161/01.STR.0000181740.74005.ee. PMID: 16151032.33. Palombo C, Kozakova M. Arterial stiffness, atherosclerosis and cardiovascular risk: Pathophysiologic mechanisms and emerging clinical indications. Vasc Pharmacol 2016;77:1–7. doi: 10.1016/j.vph.2015.11.083. PMID: 26643779.34. Schadt JC, Hasser EM. Hemodynamic effects of acute stressors in the conscious rabbit. Am J Physiol Regul Integr Comp Physiol 1998;274:814–821. doi:10.1152/ajpregu.1998.274.3.R814. PMID: 9530250.35. Lim K, Burke SL, Armitage JA, Head GA. Comparison of blood pressure and sympathetic activity of rabbits in their home cage and the laboratory environment. Exp Physiol 2012;97:1263–1271. doi: 10.1113/expphysiol. 2012.064972. PMID: 22613739.36. Guyton AC, Coleman TG. Quantitative analysis of the pathophysiology of hypertension 1969;24(5):1–19. PMID: 4306217.37. Guyton AC, Coleman TG, Cowley AW, Scheel KW, Manning RD, Norman RA. Arterial pressure regulation: Overriding dominance of the kidneys in long-term regulation and in hypertension. Am J Med 1972;52;584–594. doi:10.1016/0002-9343(72)90050-2. PMID: 4337474.38. Esler M. The sympathetic system and hypertension. Am J Hypertens 2000;13:99S–105S. doi: 10.1016/s0895-7061(00)00225-9. PMID: 10921528.39. Hall JE. The Kidney, Hypertension, and Obesity. Hypertension 2003;41:625–633. doi: 10.1161/01.HYP.0000052314.95497.78. PMID: 12623970.40. Thoren P, Jones JV. Characteristics of aortic baroreceptor C-fibres in the rabbit. Acta Physiol Scand 1977;99:448–456. doi:10.1111/J.1748-1716.1977.TB10397.X. PMID: 855672.41. Landgren S. On the excitation mechanism of the carotid baroceptors. Acta Physiol Scand 1952;26:1-34. doi.org/10.1111/j.1748-1716.1952.tb00889.x. PMID: 12985395.42. Böck P, Gorgas K. Fine structure of baroreceptor terminals in the carotid sinus of guinea pigs and mice. Cell Tissue Res 1976;170:95–112. doi: 10.1007/BF00220113. PMID: 949737.43. Hansson GK, Seifert PS, Olsson G, Bondjers G. Immunohistochemical detection of macrophages and T lymphocytes in atherosclerotic lesions of cholesterol-fed rabbits. Arterioscler Thromb 1991;11:745–750. doi: 10.1161/01. atv.11.3.745. PMID: 2029509.44. Pagano PJ, Ito Y, Tornheim K, Gallop PM, Tauber AI, Cohen RA. An NADPH oxidase superoxide-generating system in the rabbit aorta. Am J Physiol Heart Circ Physiol 1995;268:H2274–H2280. doi: 10.1152/ajpheart.1995.268.6.H2274. PMID: 7611477.45. Guo GB, Thames MD, Abboud FM. Differential baroreflex control of heart rate and vascular resistance in rabbits. Relative role of carotid, aortic, and cardiopulmonary baroreceptors. Circ Res 1982:50;554–565. doi: 10.1161/01.res.50.4.554. PMID: 6802513.46. Thames MD, Ballon BJ. Occlusive summation of carotid and aortic baroreflexes in control of renal nerve activity. Am J Physiol Heart Circ Physiol 1984;246:H851–857. doi: 10.1152/ajpheart.1984.246.6H851. PMID: 6742151.47. Hirooka Y, Imaizumi T, Sugimachi M, Takeshita A. Mechanisms involved in aortic baroreceptor excitation during drug-induced aortic pressure elevation in intact rabbits. J Auton Nerv Syst 1992;40:99–106. doi: 10.1016/0165-1838(92)90021-8. PMID: 1464698.48. Tanne JH. US guidelines say blood pressure of 120/80 mm Hg is not "normal". Brit Med J 2003;326:1104. doi: 10.1136/bmj. 326.7399.1104-a. PMID: 12763961.
Bolter CP, Turner MJ. Unmyelinated Aortic Baroreceptor Activity is Reduced in a Rabbit Model of Atherosclerosis. IgMin Res. April 29, 2026; 4(4): 141-151. IgMin ID: igmin340; DOI:10.61927/igmin340; Available at: igmin.link/p340
Anyone you share the following link with will be able to read this content:
Department of Physiology, Faculty of Biomedical Sciences, University of Otago, PO Box 56, Dunedin 9054, New Zealand
Address Correspondence:
Chris P Bolter, Department of Physiology, Faculty of Biomedical Sciences, University of Otago, PO Box 56, Dunedin 9054, New Zealand, Email: chris.bolter@otago.ac.nz
How to cite this article:
Bolter CP, Turner MJ. Unmyelinated Aortic Baroreceptor Activity is Reduced in a Rabbit Model of Atherosclerosis. IgMin Res. April 29, 2026; 4(4): 141-151. IgMin ID: igmin340; DOI:10.61927/igmin340; Available at: igmin.link/p340
Copyright: 2026 Bolter CP, et al. This is an open access article distributed under the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
![Integrated baroreceptor activity during a pressure ramp. Lines show the normalised mean activity from treated (dashed) and control (solid) animals. Representative error bars show 1SD.
(A.) Mean of normalised integrated CSN activity from 10 treated and 10 control animals. (B) Mean of normalised integrated ADN activity from 22 treated and 20 control animals. The mean area under the curve at pressures above 90 mmHg was smaller in treated than in control animals (4650 [4174, 4988] versus 5570 [5220, 5919] au; p < 0.001, single-tailed t-test.](https://www.igminresearch.es/articles/figures/igmin340/igmin340.g001.png) Figure 3.1: Integrated baroreceptor activity during a pressure...
Figure 3.1: Integrated baroreceptor activity during a pressure...
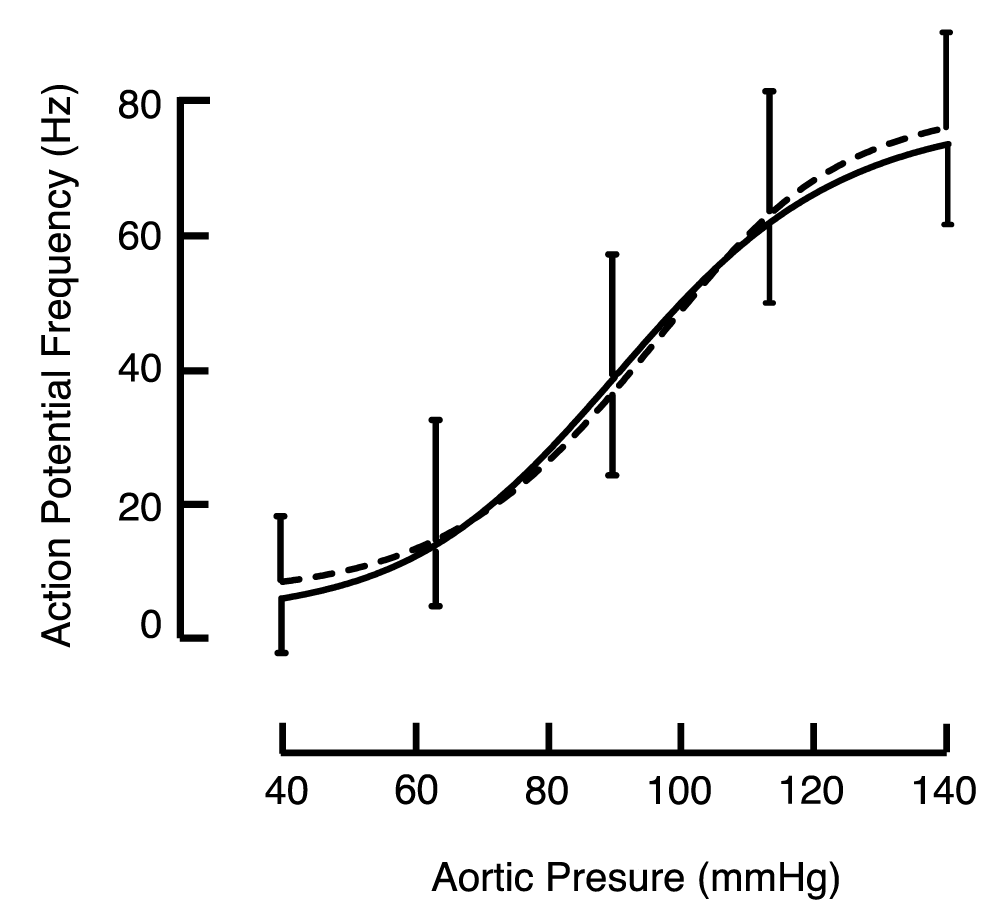 Figure 3.2: Frequency of action potentials in single barorecep...
Figure 3.2: Frequency of action potentials in single barorecep...
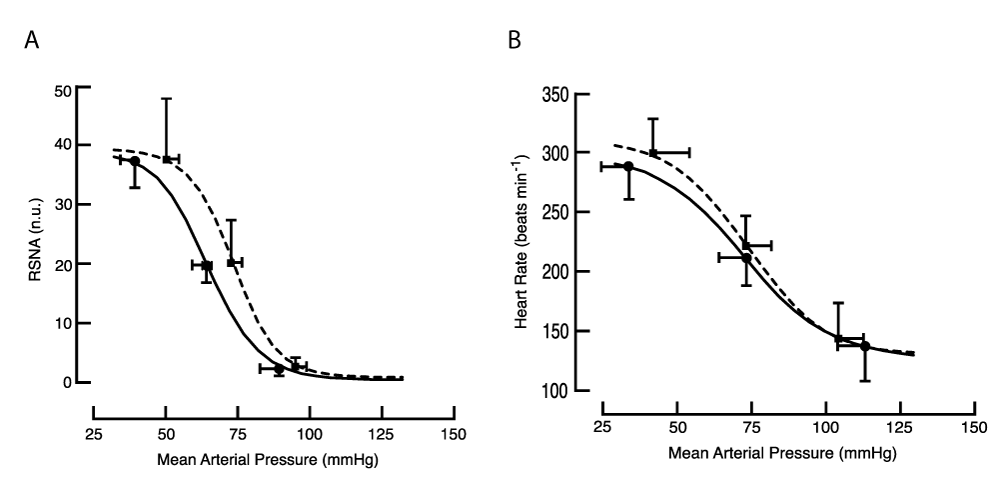 Figure 3.3: Baroreflex curves in the conscious animal. Symbols...
Figure 3.3: Baroreflex curves in the conscious animal. Symbols...
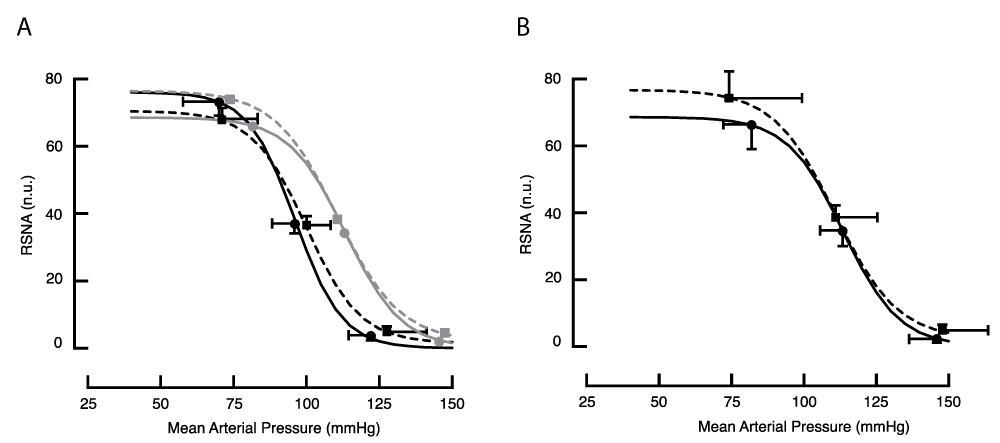 Figure 3.4: Baroreflex response curves for RSNA in the anaesth...
Figure 3.4: Baroreflex response curves for RSNA in the anaesth...
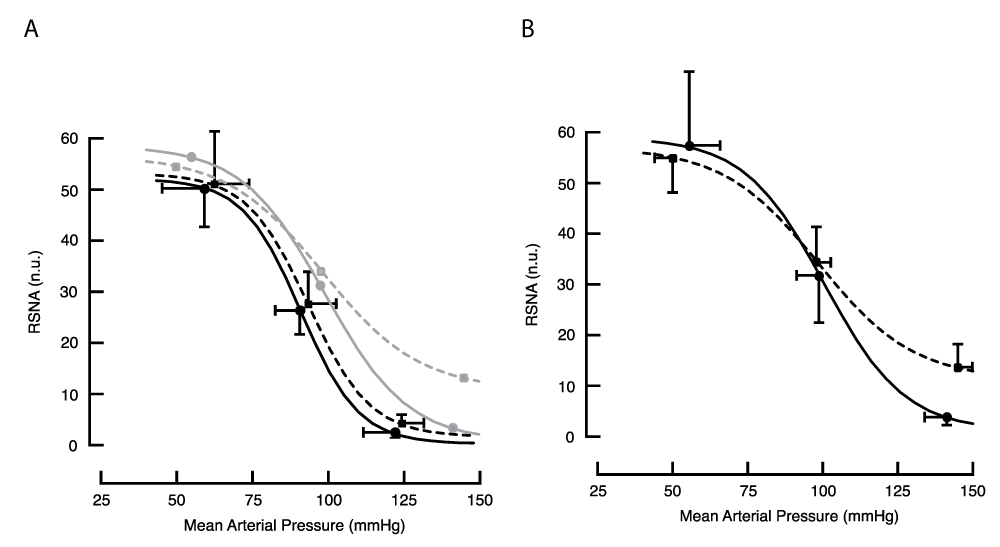 Figure 3.5: Baroreflex response curves for RSNA in the anaesth...
Figure 3.5: Baroreflex response curves for RSNA in the anaesth...
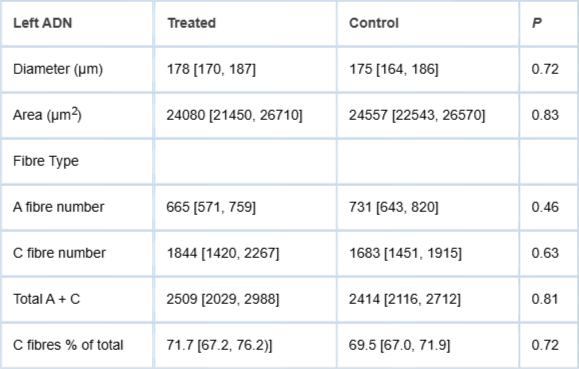 Table 3.1: Diameter, area, and fibre composition of the left ...
Table 3.1: Diameter, area, and fibre composition of the left ...
1. Incalcaterra E, Accardi G, Balistreri CR, Caimi, G, Candore G, Caruso M, et al. Pro-Inflammatory Genetic Markers of Atherosclerosis. Curr Atheroscler Rep. 2013; 15:329–337. doi: 10.1007/s11883-013-0329-5. PMID: 235916722. Jinnouchi H, Sato Y, Sakamoto A, Cornelissen A, Mori M, Kawakami R, et al. Calcium deposition within coronary atherosclerotic lesions: Implications for plaque stability. Atherosclerosis 2020; 306:85-95. doi: 10.1016/j.atherosclerosis.2020.05.017. PMID: 32654790.3. Poznyak AV, Sadykhov NK, Kartuesov AG, Borisov EE, Melnichenko AA, Grechko AV, et al. Hypertension as a risk factor for atherosclerosis: Cardiovascular risk assessment. Front Cardiovasc Med 2022; 9:959285. doi.org/10.3389/fcvm.2022.959285. PMID: 36072873.4. Libby P. The changing landscape of atherosclerosis. Nature 2021; 592:524–533. doi.org/10.1038/s41586-021-03392-8. PMID: 33883728.5. Lehmann N, Erbel R, Mahabadi AA, Kälsch H, Möhlenkamp S, Moebus S, et al. Accelerated progression of coronary artery calcification in hypertension, but also prehypertension. J Hypertens 2016; 34:2233–2242. doi: 10.1097/HJH.0000000000001080. PMID: 27512973.6. Gonzalez-Guerra A, Roche-Molina M, García-Quintáns N, Sánchez-Ramos C, Martín-Pérez D, Lytvyn M, et al. Sustained Elevated Blood Pressure Accelerates Atherosclerosis Development in a Preclinical Model of Disease. Int J Mol Sci 2021;22:8448. doi: 10.3390/ijms22168448. PMID: 34445154.7. Al-Mashhadi AL, Nasr ZP, Mortensen MB, Lewis EA, Camafeita E, Ravlo K, et al. Local Pressure Drives Low-Density Lipoprotein Accumulation and Coronary Atherosclerosis in Hypertensive Minipigs. Am Coll Cardiol 2021;77:575–589. doi: 10.1016/j.jacc.2020.11.059. PMID: 33538256.8. Mautner SL, Mautner GC, Hunsberger SA, Roberts WC. Comparison of composition of atherosclerotic plaques in saphenous veins used as aortocoronary bypass conduits with plaques in native coronary arteries in the same men. Am J Cardiol 1992;70:1380–1387. doi: 10.1016/0002-9149(92)90285-7. PMID: 1442604.9. Moore GW, Smith RR, Hutchins GM. Pulmonary artery atherosclerosis: correlation with systemic atherosclerosis and hypertensive pulmonary vascular disease. Arch Pathol Lab Med 1982;106:378–380. PMID: 6213213.10. Furukawa K, Kawano H, Futakuchi M, Ishijima M, Yamgataga Y, Ueno Y, et al. Pulmonary atherosclerosis in a patient with chronic thromboembolic pulmonary hypertension. Pathol Int 2021;71:164–166. doi:10.1111/pin. 13051. PMID: 33316138.11. Prince M, Tafur JD, White CJ. When and how should we revascularize patients with atherosclerotic renal artery stenosis? JACC Cardiovasc Interv 2019;25:505–517. doi: 10.1016/j.jcin.2018.10.023. PMID: 30898248.12. Cowley AW Jr. Long-term control of arterial blood pressure. Physiol Rev 1992;72:231–300. doi: 10.1152/physrev. 1992.72.1.231. PMID: 1731371.13. Seagard JL, Hopp FA, Drummond HA, Van Wynsberghe DM. Selective contribution of two types of carotid sinus baroreceptors to the control of blood pressure. Circ Res 1993;72:1011–1022. doi: 10.1161/01.res.72.5.1011. PMID: 8477517.14. Thrasher TN. Effects of chronic baroreceptor unloading on blood pressure in the dog. Am J Physiol Regul Integr Comp Physiol 2005:288:R863–R871. doi: 10.1152/ajpregu.00489. PMID: 15563576.15. Lohmeier TE, Barrett AM, Irwin ED . Prolonged activation of the baroreflex: a viable approach for the treatment of hypertension? Curr Hypertens Rep 2005;7:193–198. doi: 10.1007/s11906-005-0009-0. PMID: 15913493.16. Li Z, Mao HZ, Abboud FM, Chapleau MW. Oxygen-derived free radicals contribute to baroreceptor dysfunction in atherosclerotic rabbits. Circ Res 1996;79:802–811. doi: 10.1161/01. res.79.4.802. PMID: 8831504.17. Gianaros PJ, Jennings JR, Olafsson GB, Steptoe A, Sutton-Tyrrell K, Muldoon MF, et al. Greater intima-media thickness in the carotid bulb is associated with reduced baroreflex sensitivity. Am J Hypertens 2002;15:486–491. doi: 10.1016/s0895-7061(02)02923-0. PMID: 12074348.18. Tsekouras NS, Katsargyris A, Skrapar I, Bastounis EE, Georgopoulos S, Klonar C, et al. The role of carotid plaque echogenicity in baroreflex sensitivity. J Vasc Surg 2011;54:93–99. doi: 10.1016/j.jvs.2010.11.121. PMID: 21458208.19. Cox RH, Bagshaw RJ, Detweiler DK. Alterations in carotid sinus reflex control of arterial hemodynamics associated with experimental hyperlipemia in the racing greyhound. Circ Res 1980;46:237–244. doi: 10.1161/01.res.46.2.237. PMID: 7351041.20. Morita H, Nishida Y, Motochigawa H, Watanabe Y, Hosomi H. Depressed baroreflex control of renal sympathetic nerve activity in conscious WHHL rabbits. Cardiovasc Res 1988;22:678–685. doi: 10.1093/cvr/22.10.679. PMID: 2476214.21. Katsuda S, Hosomi H, Shiomi M, Watanabe Y. Impaired baroreflex control of arterial pressure in WHHL rabbits. J Vet Med Sci 1992; 54:983–987. doi: 10.1292/jvms.54.983. PMID: 1420582.22. McCully BH, Brooks VL, Andresen MC. Diet-induced obesity severely impairs myelinated aortic baroreceptor reflex responses. Am J Physiol Heart Circ Physiol 2012;302:H2083–H2091. doi: 10.1152/ajpheart.01200.2011. PMID: 22408022.23. Angell-James JE. Arterial baroreceptor activity in rabbits with experimental atherosclerosis. Circ Res 1974;40:27–39. doi: 10.1161/01.res.40.4.27. PMID: 4809347.23a. Friedewald WT, Levy RI, Fredrickson S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin Chem 1972;18:499–502. PMID: 4337382.24. Dorward PK, Burke SL, Jänig W, Cassell J. Reflex responses to baroreceptor, chemoreceptor and nociceptor inputs in single renal sympathetic neurones in the rabbit and the effects of anaesthesia on them. J Autonom Nerv Syst 1987;18:39–54. doi: 10.1016/0165-1838(87)90133–0. PMID: 3819315.25. Dorward PK, Riedel W, Burke SL, Gipps J, Korner PI. The renal sympathetic baroreflex in the rabbit. Arterial and cardiac baroreceptor influences, resetting, and the effect of anesthesia. Circ Res 1985;57:618–633. doi: 10.1161/01.res.57.4.618. PMID: 4042286.26. Burke SL, Head, GA. Method for in vivo calibration of renal sympathetic nerve activity in rabbits. J Neurosci Methods 2003;127:63–74. doi: 10.1016/s0165-0270(03)00121-3. PMID: 12865149.27. McDowall LM, Dampney RA. Calculation of threshold and saturation points of sigmoidal baroreflex function curves. Am J Physiol Heart Circ Physiol 2006;291;H2003–2007. doi: 10.1152/ajpheart.00219.2006. PMID: 16714364.28. Jones GT, Jiang F, McCormick SP, Dusting GJ. Elastic Lamina Defects Are an Early Feature of Aortic Lesions in the Apolipoprotein E Knockout Mouse. J Vasc Res 2005;42:237–246 doi: 10.1159/000085553. PMID: 15860923.29. Aumonier FJ. Histological observations on the distribution of baroreceptors in the carotid and aortic regions of the rabbit, cat and dog. Acta Anat 1972;82:1–16. doi: 10.1159/000143797. PMID: 4669378.29a. Fan J, Shimoyamada H, Sun H, Marcovina S, Honda K, and Watanabe T. Transgenic rabbits expressing human apolipoprotein(a) develop more extensive atherosclerotic lesions in response to a cholesterol-rich diet. Arterioscler Thromb Vasc Biol 2001;21:88-94. doi: 10.1161/01.atv.21.1.88. PMID: 11145938.30. Thoren P, Munch PA, Brown AM. Mechanisms for activation of aortic baroreceptor C-fibres in rabbits and rats. Acta Physiol Scand 1999;166:167–174. doi:10.1046/J.1365-201X.1999.00556.X. PMID: 10468652.31. Giannattasio C, Failla M, Emanuelli G, Grappiolo A, Boffi L, Corsi D, et al. Local effects of atherosclerotic plaque on arterial distensibility. Hypertension 2001;38:1177–1180. doi: 10.1161/hy1101.095994. PMID: 11711518.32. Makita S, Nakamura S, Hiramori, K. The association of C-reactive protein levels with carotid intima-media complex thickness and plaque formation in the general population. Stroke 2005;36:2138–2142. doi: 10.1161/01.STR.0000181740.74005.ee. PMID: 16151032.33. Palombo C, Kozakova M. Arterial stiffness, atherosclerosis and cardiovascular risk: Pathophysiologic mechanisms and emerging clinical indications. Vasc Pharmacol 2016;77:1–7. doi: 10.1016/j.vph.2015.11.083. PMID: 26643779.34. Schadt JC, Hasser EM. Hemodynamic effects of acute stressors in the conscious rabbit. Am J Physiol Regul Integr Comp Physiol 1998;274:814–821. doi:10.1152/ajpregu.1998.274.3.R814. PMID: 9530250.35. Lim K, Burke SL, Armitage JA, Head GA. Comparison of blood pressure and sympathetic activity of rabbits in their home cage and the laboratory environment. Exp Physiol 2012;97:1263–1271. doi: 10.1113/expphysiol. 2012.064972. PMID: 22613739.36. Guyton AC, Coleman TG. Quantitative analysis of the pathophysiology of hypertension 1969;24(5):1–19. PMID: 4306217.37. Guyton AC, Coleman TG, Cowley AW, Scheel KW, Manning RD, Norman RA. Arterial pressure regulation: Overriding dominance of the kidneys in long-term regulation and in hypertension. Am J Med 1972;52;584–594. doi:10.1016/0002-9343(72)90050-2. PMID: 4337474.38. Esler M. The sympathetic system and hypertension. Am J Hypertens 2000;13:99S–105S. doi: 10.1016/s0895-7061(00)00225-9. PMID: 10921528.39. Hall JE. The Kidney, Hypertension, and Obesity. Hypertension 2003;41:625–633. doi: 10.1161/01.HYP.0000052314.95497.78. PMID: 12623970.40. Thoren P, Jones JV. Characteristics of aortic baroreceptor C-fibres in the rabbit. Acta Physiol Scand 1977;99:448–456. doi:10.1111/J.1748-1716.1977.TB10397.X. PMID: 855672.41. Landgren S. On the excitation mechanism of the carotid baroceptors. Acta Physiol Scand 1952;26:1-34. doi.org/10.1111/j.1748-1716.1952.tb00889.x. PMID: 12985395.42. Böck P, Gorgas K. Fine structure of baroreceptor terminals in the carotid sinus of guinea pigs and mice. Cell Tissue Res 1976;170:95–112. doi: 10.1007/BF00220113. PMID: 949737.43. Hansson GK, Seifert PS, Olsson G, Bondjers G. Immunohistochemical detection of macrophages and T lymphocytes in atherosclerotic lesions of cholesterol-fed rabbits. Arterioscler Thromb 1991;11:745–750. doi: 10.1161/01. atv.11.3.745. PMID: 2029509.44. Pagano PJ, Ito Y, Tornheim K, Gallop PM, Tauber AI, Cohen RA. An NADPH oxidase superoxide-generating system in the rabbit aorta. Am J Physiol Heart Circ Physiol 1995;268:H2274–H2280. doi: 10.1152/ajpheart.1995.268.6.H2274. PMID: 7611477.45. Guo GB, Thames MD, Abboud FM. Differential baroreflex control of heart rate and vascular resistance in rabbits. Relative role of carotid, aortic, and cardiopulmonary baroreceptors. Circ Res 1982:50;554–565. doi: 10.1161/01.res.50.4.554. PMID: 6802513.46. Thames MD, Ballon BJ. Occlusive summation of carotid and aortic baroreflexes in control of renal nerve activity. Am J Physiol Heart Circ Physiol 1984;246:H851–857. doi: 10.1152/ajpheart.1984.246.6H851. PMID: 6742151.47. Hirooka Y, Imaizumi T, Sugimachi M, Takeshita A. Mechanisms involved in aortic baroreceptor excitation during drug-induced aortic pressure elevation in intact rabbits. J Auton Nerv Syst 1992;40:99–106. doi: 10.1016/0165-1838(92)90021-8. PMID: 1464698.48. Tanne JH. US guidelines say blood pressure of 120/80 mm Hg is not "normal". Brit Med J 2003;326:1104. doi: 10.1136/bmj. 326.7399.1104-a. PMID: 12763961.